 The visible light spectrum appears as spectral lines at 410, 434, 486, and 656 nm, which correspond to energy level transitions from n = 3, 4, 5, and 6, respectively, to n = 2. It occurs when electrons transition from an energy level higher than n = 3 back down to n = 2. The set of spectral lines in the visible light region is known as the Balmer series. This is the emission spectrum for hydrogen. With pure elemental species, the emission behavior appears as lines of specific wavelengths rather than a broad spectrum. High energy emitted light results from electrons relaxing from a higher energy level, and low energy emitted light results from electrons relaxing from a lower energy level.Īn emission spectrum is a measure of emitted radiation across a range of wavelengths. The wavelength of the absorbed and emitted light depends on the difference between the high and low energy levels. As the electrons relax down to either a lower energy state or to the ground state, the excess energy is released as a photon. The description of any electron in a multi-electron atom must find a way to characterize the effect of the other electrons on the energy.When an atom absorbs energy, the electrons become excited and move to a higher energy level. Its energy can be used to model the effective shielding as follows.Īnother way to view the energy is to say that the repulsion of the electrons contributes a positive potential energy which partially offsets the negative potential energy contributed by the attractive electric force of the nuclear charge. The fact that the second electron is less tightly bound can be interpreted as a shielding effect the other electron partly shields the second electron from the full charge of the nucleus. Since the hydrogenic energy levels depend upon the square of the nuclear charge, the energy of the remaining helium electron should be just 4x(-13.6 eV) = -54.4 eV as observed. The He+ ion is just like a hydrogen atom with two units of charge in the nucleus. The energy required to remove the second electron is 54.4 eV, as would be expected by modeling it after the hydrogen energy levels. The energy required to remove one of them is the highest ionization energy of any atom in the periodic table: 24.6 electron volts. The helium ground state consists of two identical 1s electrons. It is part of the understanding of the ordering of energy levels in multi-electron atoms. This effect is sometimes called the " spin-spin interaction" and is addressed by Hund's Rule #1. This implies that it will be more tightly bound and of lower energy. If the electrons are on the average further apart, then there will be less shielding of the nucleus by the ground state electron, and the excited state electron will therefore be more exposed to the nucleus.So in general, the probability for small separations of the two electrons is smaller than for a symmetric space wavefunction. The probability is the square of the wavefunction, and from a simple functional point of view, the square of an antisymmetric function must go to zero at the origin. An anti-symmetric space wavefunction for the two electrons implies a larger average distance between them than a symmetric function of the same type.The wavefunction for the electrons can be written as the product of the space and spin parts of the wavefunction. This forces the space part of the wavefunction to be anti-symmetric. 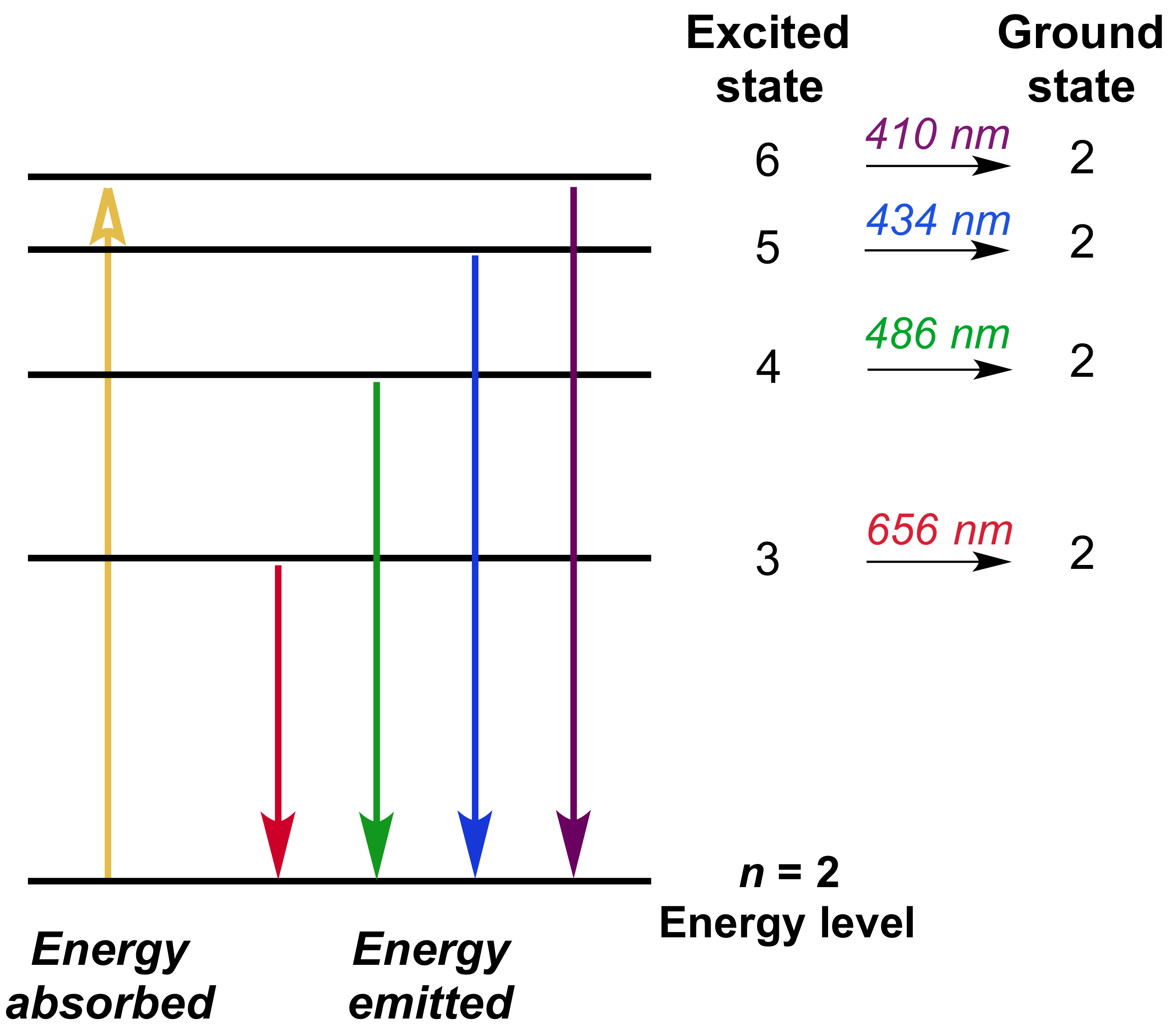
The total wavefunction for the electrons must be anti-symmetric since they are fermions and must obey the Pauli exclusion principle.The parallel spins make the spin part of the wavefunction symmetric.It is observed that the orthohelium states are lower in energy than the parahelium states. An electron in an upper state can have spin antiparallel to the ground state electron (S=0, singlet state, parahelium) or parallel to the ground state electron (S=1, triplet state, orthohelium). In the helium energy level diagram, one electron is presumed to be in the ground state of a helium atom, the 1s state. Why do levels vary with orbital quantum number? One electron is presumed to be in the ground state, the 1s state. The electron energy levels for a helium atom demonstrate a number of features of multi-electron atoms. Helium Energy Levels Helium Energy Levels 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
